Mass flow and calculations

VPInstruments flow meters measure mass flow (shown by a “n” like in mn/sec or S as in SCFM). Mass flow enables you to measure with our equipment in different pipe diameters, with different flow, pressure, or temperature ranges. Compressor output is also given in mass flow, so this makes your job easier. You can compare the results of the VPInstruments’ flow meters directly to the output of your compressor. There are a few things to take into account, though. In this article learn more about the principle of mass flow, what to be careful about and how to calculate with mass flow.
Definition of mass flow
Mass flow rate is the mass of substance which passes through a given surface per unit time. Its unit is mass divided by time. This means kilogram per second in SI units, and slug per second or pound per second in US customary units. Mass flow rate can be calculated from the density of the substance, the cross-sectional area through which the substance is flowing, and its velocity relative to the area of interest:
= ρ * v * A where: = mass flow rate
ρ = density
v = velocity
A = flow area
Relationship mass and volume in the ideal gas law
The relationship between mass, volume, temperature, and pressure for gasses is described by the ideal gas law:
P * V =n * R * T
- P = Static pressure
- V = Volume
- n = Molar mass, the amount of substance
- R = Ideal gas constant
- T = Absolute temperature

Showing the ideal gas law in a different format, directly shows the relation between volume, temperature, and pressure, as n and R are constants.
(P * V) / T = n * R
In a closed container, when pressure doubles, the volumes is divided by two at the same temperature. As can be seen in the figure to the left, the volume changes with the pressure. The number of molecules remain the same, therefore mass remains the same.
Convert mass flow to volume flow and reference conditions
People in the industry are used to flows in volume. A mass flow meter can easily provide flows in volume. How? Here is where reference conditions are important, and this makes it possible to compare different data sets.
Reference conditions define the mass of a gas, at a fixed temperature and pressure. A European reference conditions is provided at 0°C and 1.013 bar, this is called the normal reference condition. The normal reference condition is marked by an “n” as subscript in the unit, for instance mn/sec, m3n/hr.
The normal reference condition is not the only standard in the industry. E.g. the European standard reference condition is provided at 20°C and 1.013 bar. The standard reference condition is shown by a “s” as subscript in the unit, like ls/min.
Be aware that the American standard reference condition is equal to the European normal reference. This is marked as s as first letter in the unit, like SCFM. Thus, reference conditions are 20°C and 1.013 bar.
Reference conditions are vital when comparing results. Not taking these into account can provide results that are easily a few percent off, as this next example shows:
How to calculate with reference conditions?
To go from other common conditions to normal, the following formula is needed, whereby temperature is in Kelvin and pressure in mbar.
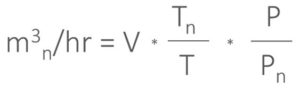
For example, to go from FAD to normal:
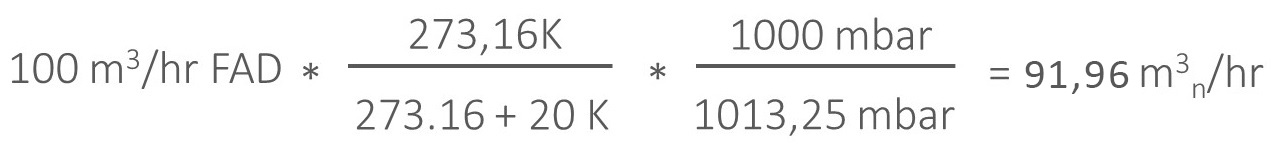

So, the difference between FAD and normal conditions is 8,7%! As compressed air is expensive, you want to take be sure your measurement output is accurate.
Advantage mass flow measurements
As this article has shown, mass flow measurements show significant advantages when measuring compressed air or gas. For correctly analyzing measurement results it is important to compare apples with apples. Always check therefore the measurement unit, are you measuring volumetric flow or mass flow. And when comparing mass flow measurements, make sure to check the reference conditions.

VPFlowScope and mass flow
The VPFlowScope flow meters are calibrated in normal meters per second (mn/sec). A normal meter per second is a meter per second under the same reference conditions as a normal cubic meter. This enables you to use the VPFlowScope in various pipe diameters. The VPFlowScope only needs to know the inner tube diameter to calculate the mass flow rate.














